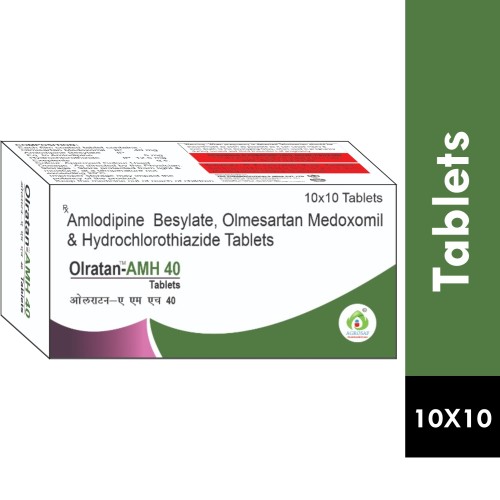
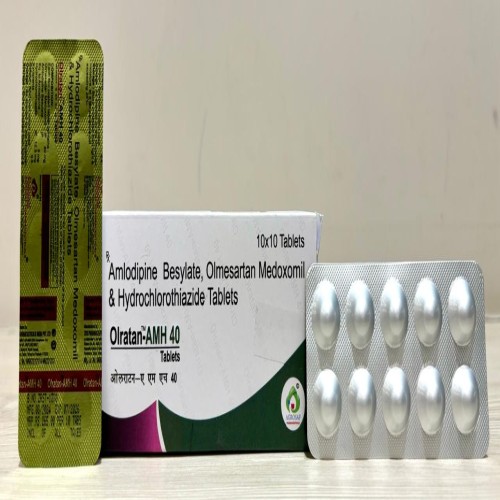
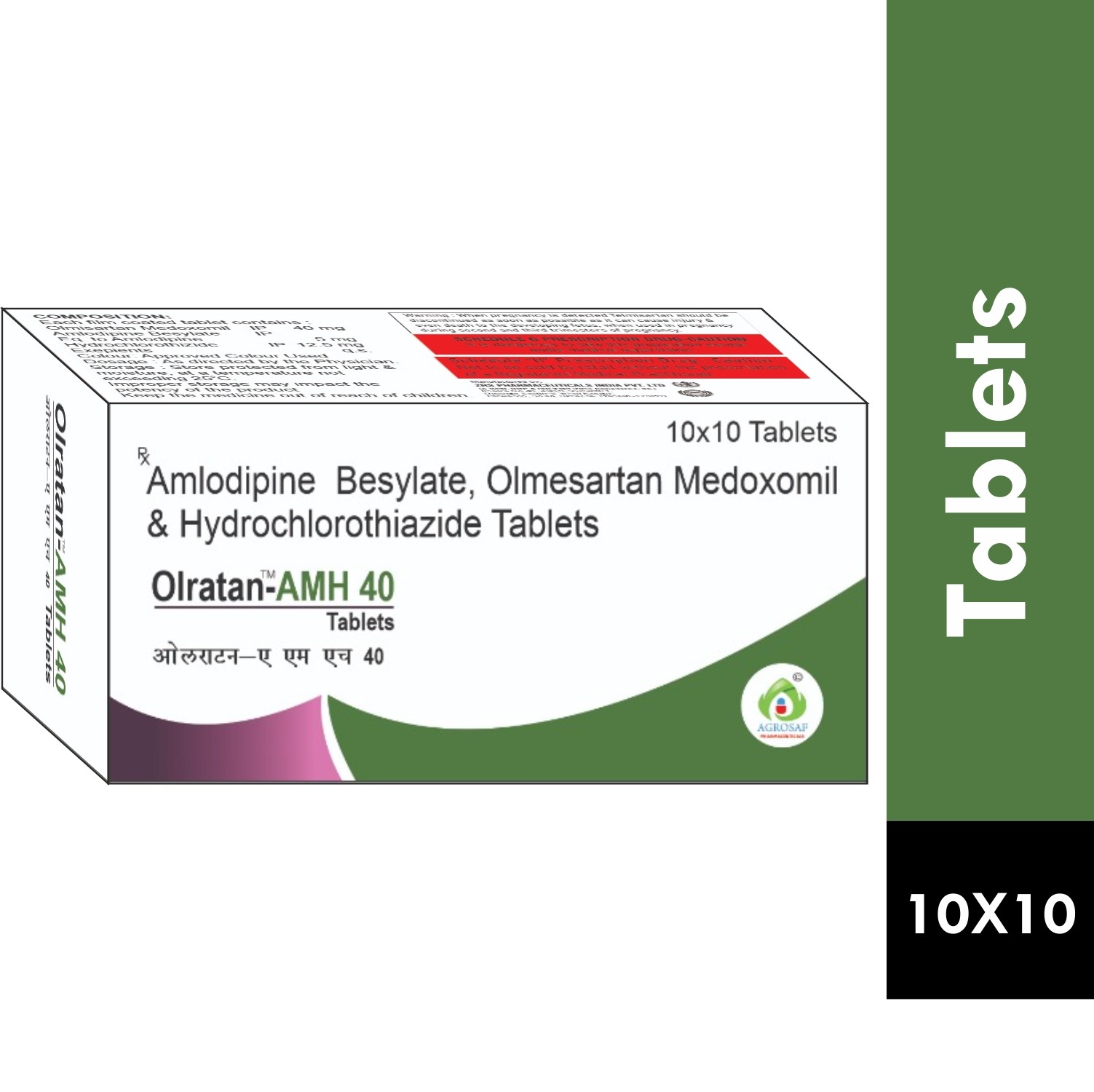
| PACKAGING TYPE: | 10x10 Alu-alu (10 Strips In 1 Box) |
| COMPOSITION: | Olmesartan Medoxomil(40mg),amlodipine(5mg) & Hydrochlorothiazide(12.5mg) Tablet |
| FORMULATION: | Tablet |
| Description: | OLRATAN AMH 40 Tablet contains a combination of Olmesartan Medoxomil 40 mg, Amlodipine 5 mg, and Hydrochlorothiazide 12.5 mg. This triple-action antihypertensive formulation is designed for patients requiring stronger blood pressure control. Olmesartan relaxes blood vessels by blocking angiotensin II, Amlodipine dilates arteries to reduce vascular resistance, and Hydrochlorothiazide promotes sodium and water excretion, helping to manage fluid retention and providing comprehensive cardiovascular protection. |
| Side Effects: | Common: Dizziness or lightheadedness Swelling of feet or ankles (edema) Headache Fatigue Flushing Increased urination Serious (Rare): Severe hypotension Kidney function changes Electrolyte imbalance (low potassium or sodium) Allergic reactions (rash, swelling, difficulty breathing) |
| How to Use: | Take as prescribed by a healthcare professional Usually once daily, with or without food Swallow the tablet whole with water Do not double doses if missed; take the next dose as scheduled |
| Precaution: | Monitor blood pressure, kidney function, and electrolytes regularly Use with caution in patients with liver or kidney disease Not recommended during pregnancy Inform your doctor about other medications being taken Avoid potassium supplements unless prescribed |
| Product Benefits: | Provides enhanced blood pressure control through triple mechanism Reduces risk of stroke, heart attack, and kidney complications Helps manage fluid retention and improves cardiovascular health Suitable for patients not adequately controlled with monoth |
Frequently Asked Questions
It is used for high blood pressure management, especially in patients requiring triple therapy for stronger blood pressure control.
Yes, mild dizziness, swelling (edema), and electrolyte imbalances are possible. Regular monitoring is recommended.
Yes, under medical supervision with regular monitoring of blood pressure, kidney function, and cardiovascular health.